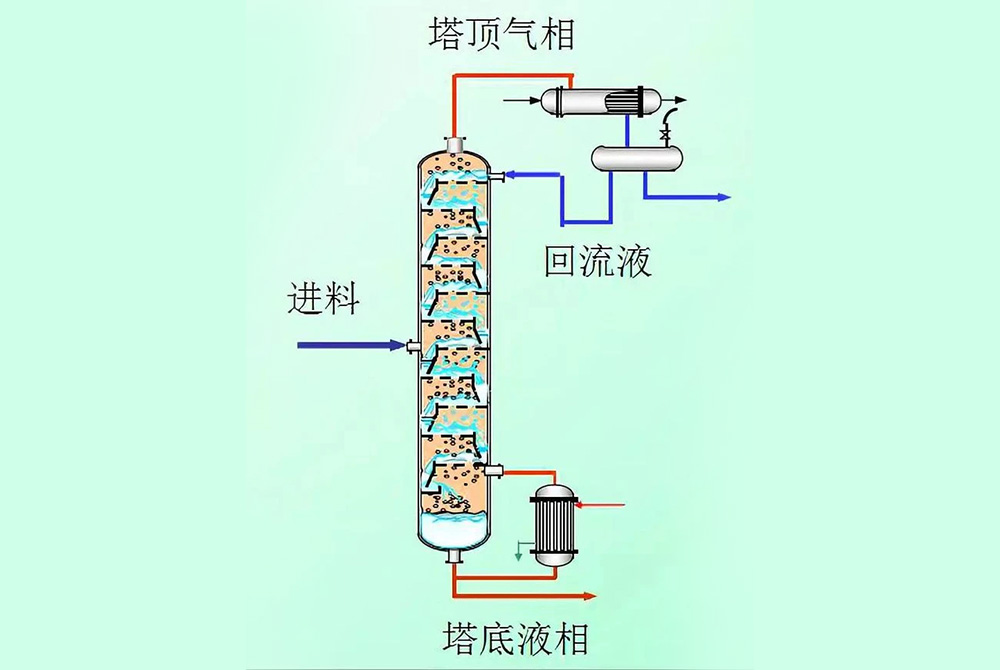
Distillation tower solvent separation
Overview: Distillation is a thermodynamic separation process that involves heating a mixture of liquids to evaporate the lower boiling components into steam, which is then condensed back into a liquid state to achieve the separation of different boiling point components in the mixture. Simply put, distillation is a technique that utilizes the difference in boiling points between components to separate liquid mixtures.
Distillation is a more complex and efficient form of distillation, mainly used to separate liquid mixtures with similar boiling points. The distillation process is carried out in a special equipment - a distillation tower, which is equipped with trays or packing inside the tower, allowing the rising steam to come into contact and transfer mass with the falling condensate multiple times, thereby achieving slight evaporation and condensation at each stage, gradually improving the purity of the product. In short, distillation achieves a higher degree of separation and purification through a continuous process of multiple partial evaporation and condensation. Below, we will explain the principle of distillation in two ways.
Distillation principle and steps:
1. Heating mixture: First, place a mixture containing multiple volatile components into a closed container (usually referred to as a distillation flask or flask) and heat it. The purpose of heating is to convert the lower boiling components in the mixture into steam. The temperature should be controlled below the boiling point of the lowest boiling component in the mixture to avoid unnecessary decomposition or reaction.
2. Generating steam: As the temperature increases, the component with the lowest boiling point in the mixture begins to evaporate and form steam. Steam mainly contains components with lower boiling points, while components with higher boiling points remain in liquid form.
3. Steam condensation: The steam is then directed to the condenser, where it condenses back into a liquid state due to cooling. The design of the condenser allows steam to fully contact the cooling surface, effectively transforming into liquid droplets. The ideal condenser can ensure that almost all steam is converted back to liquid.
4. Collecting fractions: The condensed liquid, also known as fractions, is collected in a receiving container. In simple distillation, the first fraction collected usually has the lowest boiling point, and as the distillation process progresses, the boiling points of subsequent fractions gradually increase. This method of sequential collection is called "fractionation".
5. Repeated distillation: For components with boiling points very close, simple distillation may not achieve sufficient separation effect. At this point, more complex distillation methods may need to be used, such as fractional distillation or vacuum distillation. The fractionation column is equipped with fillers or trays, which can provide a large surface area to promote heat exchange between steam and condensate, achieving more efficient separation.
6. Refining and Purification: Through repeated distillation processes, products can be further purified, especially for applications that require extremely high purity, such as laboratory preparation of pure reagents or industrial production of high-purity chemicals.
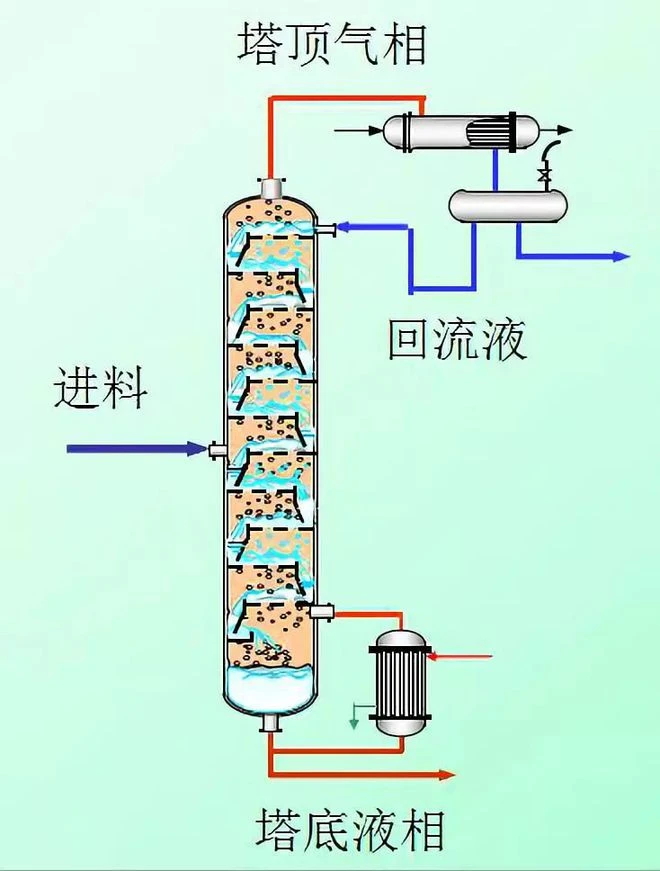
Using distilled spirits as an example, explain the principle of distillation in a simple way:
1. Mixed liquid heating: We have a bucket of mixed liquid containing water and different concentrations of alcohol, and we want to extract higher purity alcohol from it. Firstly, like steaming wine, we heat this barrel of mixed liquor. As the temperature rises, alcohol (ethanol) and water begin to evaporate because their boiling points are relatively close (alcohol at around 78 ° C, water at 100 ° C), but there are still differences.
2. Entering the distillation tower: The steam generated by evaporation is directed into a device called the distillation tower. This tower has many floors, like multiple staircases in a building. As steam rises, it encounters colder surfaces on each level. At this point, water with a higher boiling point is more likely to condense back into a liquid state than alcohol and "stay" on the lower floors of the tower, while alcohol vapor continues to rise.
3. Step by step separation: As the steam continues to rise and each layer condenses and refluxes, the low boiling point alcohol vapor gradually concentrates in the upper part of the tower, while the high boiling point water and other impurities with boiling points higher than alcohol accumulate in the lower layers. In this way, the distillation tower acts as an efficient sorting machine, gradually separating increasingly pure alcohol vapor.
4. Top collection: Ultimately, the purest alcohol vapor reaches the top of the tower, where a condenser awaits it to transform it into liquid form - this is high-purity alcohol, which can be collected directly. This process can be repeated, and each cycle can further improve the purity of the alcohol.
5. Continuous optimization: In practical operation, precise control of temperature, pressure, and reflux ratio (the proportion of a portion of the condensate returning to the bottom of the tower for reheating and evaporation) will be used to optimize the separation effect and ensure the highest quality of alcohol products.
In short, the distillation principle of alcohol purification is the process of accurately controlling heating, evaporation, and multi-stage separation to gradually and efficiently separate alcohol, water, and other impurities using boiling point differences, ultimately obtaining high-purity alcohol.
Category:
Distillation can be classified into various categories based on different operating principles, equipment structures, and application requirements. Here are some of the main distillation classifications:
1. According to the operating pressure:
Atmospheric distillation: a distillation process carried out under conditions close to atmospheric pressure.
Pressure distillation: Operated at pressures higher than atmospheric pressure, suitable for separating mixtures with high boiling points that are difficult to distill at atmospheric pressure.
Vacuum distillation: carried out under conditions lower than atmospheric pressure, particularly suitable for the separation of thermosensitive substances, which can lower the operating temperature and prevent substance decomposition or deterioration.
2. According to the tray structure
Plate tower distillation: Multiple layers of trays are installed inside the tower, and gas-liquid two-phase mass and heat transfer occur on the trays.
Packing tower distillation: using packing instead of tray, gas and liquid come into countercurrent contact between packing, common packing includes Raschig ring, Bauer ring, etc.
3. According to the operation method
Continuous distillation: Continuous addition of raw materials and continuous extraction of products, suitable for large-scale industrial production.
Intermittent distillation: adding raw materials at once, distilling in batches until separation is complete, commonly used in small-scale experiments or special product production.
4. According to special design:
Extraction distillation: Adding a third component (extractant) to the traditional distillation system to achieve effective separation of difficult to separate components by changing the relative volatility of the system.
Constant boiling distillation: For systems that form constant boiling compounds, a new constant boiling compound is formed by adding a third component, which changes the interaction between the original components and achieves separation.
Multi effect distillation: By connecting multiple distillation towers in series, the top steam of the previous tower serves as the heat source for the subsequent tower, achieving energy savings.
Application areas of distillation:
Very extensive, covering multiple industries such as chemical industry, petroleum processing, pharmaceuticals, food and beverage, environmental engineering, etc
1. Petrochemical industry: Various hydrocarbon mixtures produced by petroleum cracking are separated into high-purity single compounds through distillation, such as gasoline, diesel, kerosene, ethylene, propylene, etc.
2. Chemical industry: Distillation is used in the production of organic chemicals to purify various organic solvents such as alcohols, ketones, acids, esters, as well as monomers for synthetic materials, such as vinyl chloride (PVC) raw material.
3. Pharmaceutical industry: Distillation is used in the drug manufacturing process to purify raw materials, intermediates, and solvents, ensuring that drug quality meets high standards, such as the purification of antibiotics and vitamins.
4. Alcoholic beverage production: Ethanol is extracted and highly concentrated from fermentation broth through distillation, and is used to produce strong spirits such as vodka, whiskey, etc. It is also used in the manufacture of alcohol disinfectants.
5. Air separation: Large scale air separation units in industry use distillation technology to separate gases such as oxygen, nitrogen, and argon from the air, serving fields such as healthcare, metal smelting, and food preservation.
6. Environmental Protection and Waste Recycling: Distillation technology is used in wastewater treatment and exhaust gas purification to recover valuable chemicals while reducing environmental pollution, such as solvent recovery.
7. Natural product extraction: Distillation is used in the spice, cosmetics, and health product industries to separate high-value aromatic compounds and active ingredients from plant extracts, such as rose essential oil and peppermint oil.

Physical picture of Hanchuang distillation tower case