Overview: Distillation is a thermodynamic separation process that involves heating a mixture of liquids to evaporate the lower boiling components into steam, which is then condensed back into a liquid state to achieve the separation of different boiling point components in the mixture. Simply put, distillation is a technique that utilizes the difference in boiling points between components to separate liquid mixtures.
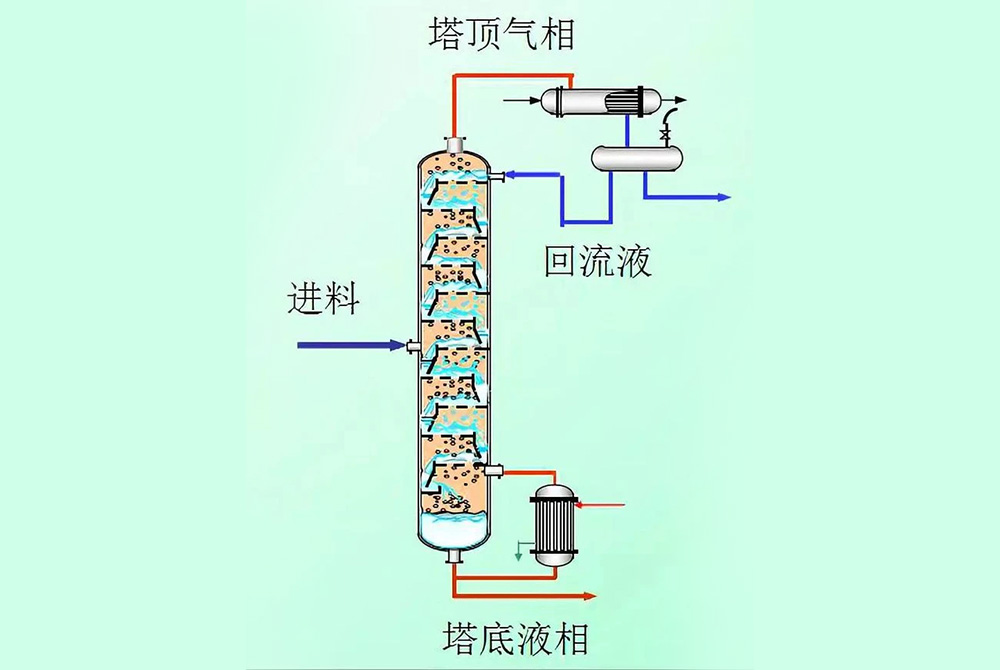
Distillation is a more complex and efficient form of distillation, mainly used to separate liquid mixtures with similar boiling points. The distillation process is carried out in a special equipment - a distillation tower, which is equipped with trays or packing inside the tower, allowing the rising steam to come into contact and transfer mass with the falling condensate multiple times, thereby achieving slight evaporation and condensation at each stage, gradually improving the purity of the product. In short, distillation achieves a higher degree of separation and purification through a continuous process of multiple partial evaporation and condensation. Below, we will explain the principle of distillation in two ways.