Catalytic Combustion
Overview: Catalytic combustion is a typical gas-solid phase catalytic reaction, which essentially involves the participation of reactive oxygen species in deep oxidation. In the process of catalytic combustion, the role of the catalyst is to reduce the activation energy and enrich the reactant molecules on the surface, thereby increasing the reaction rate. With the help of catalysts, organic waste gas can undergo flameless combustion under lower ignition temperature conditions, and be oxidized and decomposed into CO2 and H2O, while releasing a large amount of heat energy. The reaction process is as follows:
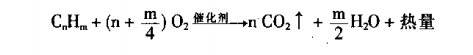
1、Characteristics of catalytic combustion
1. Low ignition temperature. Energy saving. Compared with direct combustion, catalytic combustion of organic waste gas has significant characteristics of low ignition temperature and low energy consumption. In some cases, no external heating is required after reaching the ignition temperature
2. Widely applicable. Catalytic combustion can treat almost all hydrocarbon organic waste gases and odorous gases. For low concentration, multi-component, and non recyclable waste gases emitted from industries such as organic chemicals, coatings, and insulation materials, the adsorption catalytic combustion method has a better treatment effect.
3. High processing efficiency and no secondary pollution. The purification rate of organic waste gas treated by catalytic combustion method is generally above 95%, and the final products are harmless CO2 and H2O, so there is no secondary pollution problem. In addition, due to the low temperature, it can significantly reduce the generation of NOx.
2、Composition and activity of catalyst
1. Catalysts and carriers
Catalysts are usually composed of active ingredients, additives, and carriers. The active components of catalytic reactions are loaded on carriers with larger specific surface areas. In catalytic reactions, the role of the carrier is not only to load and disperse the active components, but also to increase the stability, selectivity, and activity of the catalyst. So, the activity of a catalyst depends not only on the distribution of its active components, but also on the size and chemical state of the carrier particles, that is, the catalyst carrier has a significant impact on the catalytic effect and lifespan. The active components of catalysts can be divided into precious metal and non precious metal oxides: precious metals Pt and Pd are the most commonly used catalysts for low-temperature catalytic combustion, with the advantages of high catalytic activity and good toxicity resistance. The disadvantages are that the active components are prone to volatilization and sintering, followed by high prices; Non precious metal catalysts mainly include oxides of transition metals such as copper, manganese, and chromium, as well as perovskite type composite oxide catalysts. They have relatively low prices and good high-temperature stability, but their disadvantages are relatively low catalytic activity and high ignition temperature requirements.
2. There are three main types of catalyst poisoning and their prevention and control
(1) The catalyst is completely poisoned. Toxic substances form strong binding forces with the active centers of catalysts, which cannot be removed by conventional methods or cannot be removed at all.
(2) Inhibit catalytic reactions. Compounds of halogens and sulfur are prone to binding with active centers, and this action is reversible.
(3) Sedimentation covers active centers. Carbon deposition, ceramic dust, and other particulate matter caused by combustion block the active centers, affecting the adsorption capacity of the catalyst and reducing its activity.
3、Catalytic combustion process flow
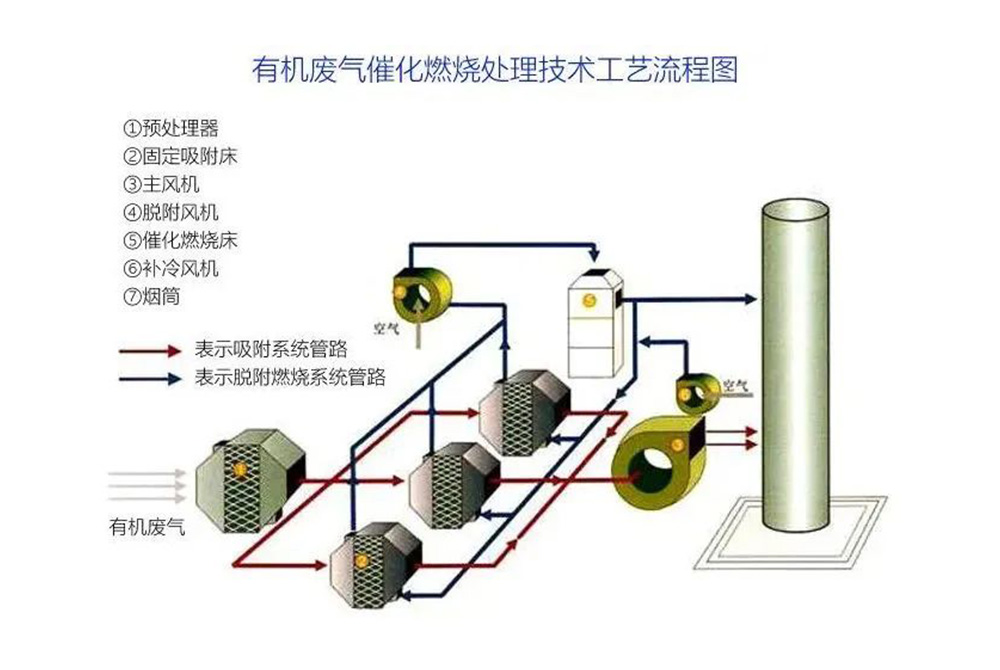
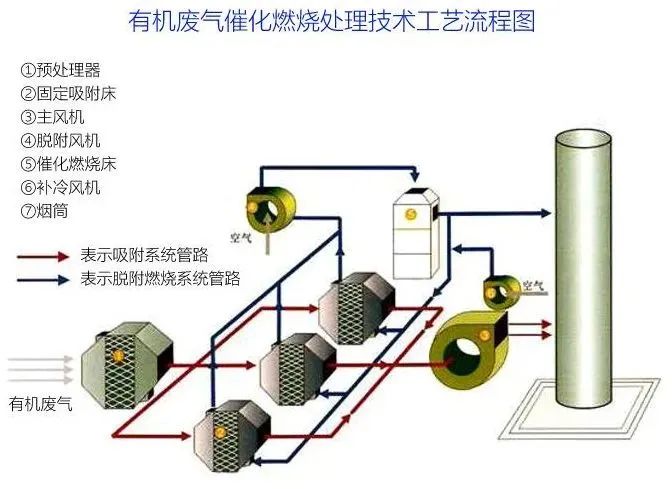
According to the preheating and enrichment methods of exhaust gas, the catalytic combustion process can be divided into three types: preheating, natural thermal equilibrium, and adsorption catalytic combustion.
The measures to reduce the decay of catalyst activity are as follows: accurately control the reaction conditions according to the operating procedures; Pre treat exhaust gas to prevent catalyst poisoning; When carbon deposits on the surface of the catalyst, excessive air is blown in and the combustion temperature is increased to remove the surface carbon deposits; Improve the preparation process of catalysts, enhance their heat resistance and anti toxicity ability.
4、Catalytic combustion process flow
According to the preheating and enrichment methods of exhaust gas, the catalytic combustion process can be divided into three types: preheating, natural thermal equilibrium, and adsorption catalytic combustion.
1. Preheating type
This is the most basic process form of catalytic combustion (see Figure 1). The temperature of organic waste gas is below 100 ℃, and the concentration is also low, so the heat cannot be self-sufficient. Therefore, it needs to be heated in the preheating chamber before entering the reactor. After combustion purification, the gas exchanges heat with untreated exhaust gas in the heat exchanger to recover some heat. This process usually uses gas or electric heating to raise the temperature to the ignition temperature required for catalytic reactions.
2. Self thermal equilibrium type
When the discharge temperature of organic waste gas is higher than the ignition temperature (around 300 ℃) and the organic content is high, the heat exchanger recovers some of the heat generated by the purified gas, which can maintain thermal balance under normal operation without the need for additional heat. Usually, only an electric heater needs to be installed in the catalytic combustion reactor for use during ignition.
3. Adsorption catalytic combustion
When the flow rate of organic waste gas is high, the concentration is low, the temperature is low, and a large amount of fuel is required for catalytic combustion, adsorption can be used to first adsorb the organic waste gas onto adsorbent t for concentration, and then hot air can be blown to desorb the organic waste gas into concentrated high concentration organic waste gas (which can be concentrated more than 10 times), and then catalytic combustion can be carried out. At this point, there is no need to supplement the heat source to maintain normal operation.
5、Application of Catalytic Combustion
1. Application of Catalytic Combustion in Automotive Exhaust Purification
Catalytic purification of automobile exhaust is the process of converting CO, CH, and NOx from the exhaust pipe into CO2, H2O, and N2 through catalytic reactions using a catalytic converter installed at the rear of the vehicle. This catalyst is a three-way catalyst. To prevent catalyst poisoning, it is necessary to use fuel with low sulfur and lead content. The development of catalysts with in-situ NOx reduction ability in the presence of a large amount of excess oxygen can achieve a 25% fuel savings in automotive engines. With the application of new materials and the promotion of low sulfur gasoline, the market prospect of catalytic combustion technology is broad.
2. Application of catalytic combustion in the treatment of dioxin gas
Catalytic combustion technology is used to treat dioxin gas at 240 ℃ -260 ℃ and 8000r? At the speed of H-1, the removal rate of dioxins reaches 99%, and the concentration of dioxins can be reduced to below 0.1ng/m3. The precursor substances of dioxins such as polychlorinated biphenyls and furans in exhaust gas are basically completely decomposed, and nitrogen oxides undergo selective catalytic reduction reactions to generate harmless nitrogen gas.
3. Application of Catalytic Combustion in Treating Industrial Organic Waste Gas
The use and emission of organic volatile compounds are involved in industrial production processes such as petrochemicals, paints, coatings, and tire manufacturing. If these volatile organic compounds (VOCs) are not treated and directly discharged into the atmosphere, they will cause serious pollution to the environment. Catalytic combustion purification technology converts organic molecules into harmless carbon dioxide and water through deep oxidation on the surface of the catalyst. Catalytic combustion technology has transitioned from experimentation to engineering practice and is gradually being applied to the purification and treatment of organic waste gases in industries such as petrochemicals, pesticides, printing, and coatings.